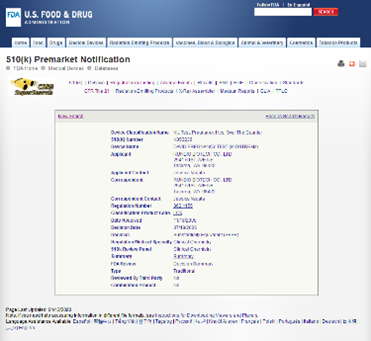
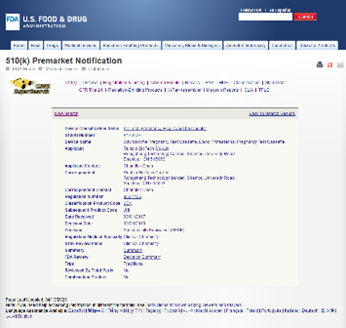
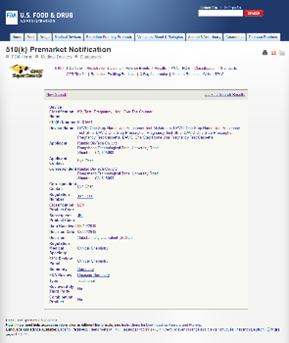
Equinox established a quality management system in June 2007, obtained the ISO13485:2003 QMS certificates from TUV in July 2010 and successfully passed the annual audit from then on. Besides, our QMS also meets United States 21 CFR 820 requirements. Our facility has been registered in the United States and passed relevant inspection in September 2010. Beyond that our QMS also meets relevant requirements of MDSAP regulation and obtained MDSAP QMS certificate from TUV in 2018, and successfully passed the annual audit from then on. With the support of the well-established quality management system, we obtained CE certificate, 510 (k) approval and certificates issued by Health Canada for self-testing products.